Paris, will open in May (18-19) with connectivity, sustainability and user experience identified as the key trends in 2022 influencing innovation across pharmaceutical packaging and drug delivery.
The event – which unites major players in pharmaceutical packaging, drug delivery, medical devices and machinery – returns to the Paris Expo, Porte de Versailles (Hall 7.2) and will see 5000 attendees and some 300+ companies exploring next generation technologies and looking ahead to debate the major industry developments and growth opportunities.
Pharmapack Europe will be held as Smart Event to enhance the benefits for global attendees, providing a hybrid experience that extends the value of Pharmapack beyond the two days in Paris. One week prior to the event, and for one week post, attendees will be able to access a dedicated online platform with bespoke content, analysis and networking. This gives additional opportunities to connect and create meetings outside of the event, increasing the number of companies attendees can meet with, while maximising invaluable ‘at event’ time.
The agenda, in response to industry needs, will span three core tracks: Design & Development, Sustainability, and Combination Products & Biologics. Speakers include a ‘who’s who’ of packaging and delivery heavyweights – with market analysts, regulatory experts and executives from some of the sector’s most innovative companies and emerging start-ups.
“As we emerge out of the pandemic, we are seeing a significant number of opportunities for drug delivery and packaging companies. The key focus for industry is shifting towards more connected devices, improving the overall patient experience and, of course, how we can overcome the next big challenge in ensuring pharma is a more sustainable industry. What has underpinned these positive developments is more diverse partnering and sharing of insights – which is why we have spent the last few months aligning the event with everything the industry needs to prosper,” commentedSherma Ellis-Daal, Brand Manager at Pharmapack Europe.
The Design & Development Track features insights from device guru, Thomas Watts, Engineering Consultant at Team Consulting, who explores the Challenges of Cloud Connectivity. In particular, he will discuss how innovators can avoid many of the common pitfalls faced when integrating connectivity into novel devices.
Two hot areas likely to significantly impact all industry thinking in the next year are how to make packaging both more eco and user-friendly. Eco design analysis will be provided by speaker Romane Osadnick – Key Account Manager, Adelphe – as he explores how to comply with regulatory requirements for the application of Eco-Design in Pharmaceutical Packaging. From the consumer sector’s perspective, Clare Beddoes, Senior Insights Research Consultant, Cambridge Design Partnership and Ben Kubler, Consultant Industrial Designer, Cambridge Design Partnership will breakdown the current landscape on how Drug Delivery is Evolving the User Experience.
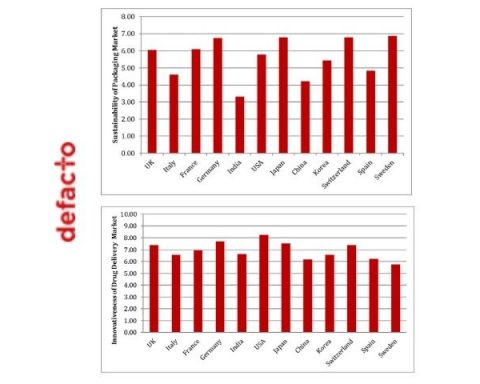
In contrast, the highly anticipated second track, will delve into the defining challenge of the next decade, exploring sustainability implications for devices and packaging. Tom Oakley, Director of Drug Delivery Device Development, Springboard, will speak on the Strategies for Environmental Sustainability – looking in particular at thetransition we are seeing to sustainability being seen as a primary concern for the drug delivery device industry.
Another environmental concern gaining increased traction is the medical waste generated at healthcare facilities. Most excitingly new innovations are now helping to overcomes this challenge. Mayur Patel, Digital Health Expert, PA Consulting explores the Duality of Safety and Efficacy in Green Drug Delivery. His session demonstrates how your company can join others leading ‘the economy of the future’ by focusing on minimizing waste and maximizing resources through reuse, distribution, and recycling.
The third track is entitled, Combination Products & Biologics, with four sessions featuring topics on packaging materials and regulations. One eagerly anticipated area is the future impact of mRNA technologies after the ground-breaking success of the two Covid vaccines. Tom Van Ginneken, Head of Product Management Polymer Solutions, SCHOTT AG will discuss the Long-Term Effects of mRNA Delivery, exploring the packaging considerations of such low temperature products.
In his insightful session on regulatory strategies, Fabien Roy, Partner, Hogan Lovells International LLP, will review the process and, considerations of, registering innovations so that they clear regulatory review and approval smoothly.
Finally, Asmita Khanolkar, Senior Director, Cambridge Pharma, SMC will discuss integrated approaches for the rapid development of novel therapies and the Dynamics and Outlook on High-Volume Subcutaneous Drug Delivery. Peter J. Schmitt, Managing Director & Co-Founder, Montesino will discuss The Limitations of Incumbent Materials. Exploring the intertwined challenges of Extractables and Leachables, and whether incumbent materials can meet the challenge or if it is time they are replaced by engineering grade polymers.
“Our events are now the tip of a 365 approach to industry partnering. Everything we do is about accelerating partnering and innovation process. So, in addition to the conference, we will be running a 2-day Symposium, Learning Lab presentations, the eponymous Awards ceremonies, as well as Start-up Hub, Innovation Gallery and tours,” added Ellis-Daal.
The 17 learning labs are a collection of short presentations on the latest drug delivery and device innovations, with highlights including, Aptar Pharma’s presentation on the ‘benefits of nasal drug delivery’ and West’s session on ‘selecting optimal pre-filled syringe components for Covid 19 vaccines’.
Additionally, the highly anticipated Pharmapack Report 2022 will be released at Pharmapack Europe, and is open for industry survey responses. To complete the survey and receive a pre-release copy of the findings, please visit: https://www.surveymonkey.co.uk/r/JWL5QB7